Even “never-smoking” U.S. vapers mostly used nicotine products
15th November 2016 - News analysis |
Even the tiny proportion of e-cig users who have never been smokers have mostly used other nicotine products, according to a study of U.S. vapers.
Celebrating our 10th anniversary – Unlock our special offer today
Even the tiny proportion of e-cig users who have never been smokers have mostly used other nicotine products, according to a study of U.S. vapers.
The Welsh are attempting to pass the Public Health Bill once again. This time minus a ban on public vaping.
Most non-vaping Americans would not permit someone else to use an e-cigarette in their house, research suggests.
Opponents of animal testing are worried that the U.S. deeming regulations for e-cigarettes will lead to the industry conducting more scientific research on live animals.
As the third day of the World Health Organization’s seventh Conference of the Parties (COP7) came to a close in India, the WHO was busy pushing for all loopholes to be closed in article 5.3 of the Framework Convention on Tobacco Control (FCTC).
E-cigarette products containing nicotine cannot be sold in Turkey without a pharmaceutical licence from the Ministry of Health – and so far no such licence has been awarded.
Amid protests from local farmers, the seventh session of the Conference of Parties is underway in New Delhi with a focus on the illegal tobacco trade and Electronic nicotine delivery systems (ENDS) – which the WHO has been critical of.
A growing number of reported e-cigarette battery incidents is leaving the industry facing a serious problem despite the low statistical probability of an event happening.
Young Americans are becoming more confident that vaping is safer and less addictive than smoking, researchers have found – but among older smokers the perception is moving in the opposite direction.
A negative report on e-cigarettes presented by the World Health Organization to the seventh session of the tobacco control conference in New Delhi has been scathingly criticised by the UK Centre for Tobacco and Alcohol Studies, which accuses the authors of the brief report of “naïvety”.
The French government has endorsed the use of e-cigarettes for smoking cessation after a meeting with pro-vaping associations.
We strongly expect nicotine-containing e-cigarettes to be legalised in Norway, where their sale has been banned until now. The anticipated move will come as a boost to the Norwegian vaping market, which has already grown by 18% since 2014.
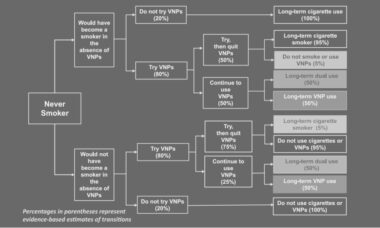
New data from the U.S. Centers for Disease Control and Prevention (CDC) seems to indicate that there are high levels of dual use amongst U.S. vapers and a significant percentage of vapers that have never smoked conventional cigarettes.
Muddled definitions of addiction are undermining the recognition of e-cigarettes as reduced-risk products, according to specialists in harm reduction.
Nearly a third of Russian high-school students have tried vaping, according to new research.
The U.S. Food and Drug Administration (FDA) will soon start requiring e-cig manufacturers to supply it with documentation on health-related issues.
This report examines the regulation of heat-not-burn (HnB) or heated tobacco products in Europe, as well as some of the broader regulatory issues that they raise. It considers their treatment under the TPD; the question of whether they are smoking or smokeless products; and issues such as labelling, advertising and taxation. A detailed case study focuses on Italy.
The U.S. Food and Drug Administration (FDA) has confirmed that some unapproved e-cigarettes can be used in scientific research as long as their makers meet the deadlines for compliance.
Italy has adopted a light touch approach to implementing the European TPD regulations, with only minimum restrictions on the e-cigarette industry. There is only a limited ban on advertising, and no restriction on public use of e-cigs. Cross-border sales are prohibited, but domestic distance sales are allowed. Our report provides a full examination of all aspects of post-TPD e-cigarette regulation in Italy.
Regulation of vape products in the Netherlands follows closely the provisions of the European Tobacco Products Directive (TPD). But although the vaping industry is highly regulated, a few issues remain to be resolved, such as what laboratories will be accredited for product testing. This complete regulatory report examines in detail every aspect of e-cigarette regulation in the Netherlands.
Recent news reports have lambasted hoverboards over safety concerns – prompting multiple retailers to drop listings. But is the threat over-rated? There are some striking similarities to similar news reports on e-cigarettes.
Following the introduction this summer of the deeming regulations by the U.S. Food and Drug Administration (FDA), each vapour product manufacturer or importer is required to submit health documents describing the physiological, behavioural and toxicological effects of their tobacco products, current or future.
Most e-cigarette smokers are willing to vape in areas where it is forbidden to smoke, U.S. research suggests.
State legislatures in the U.S. continue to have numerous e-cigarette-related bills placed before them. Totting up the topics of those proposed laws reveals that some issues remain major concerns at state level, while others seemingly don’t trouble the politicians much.
Scientists and researchers have said that anti-smoking charities are acting like the anti-Communist zealots of the 1950s McCarthy era in trying to keep them away from an international conference on tobacco and nicotine. One public-health expert said: “The tactic is to say ‘tobacco industry’ like senator Joseph McCarthy would once say ‘communist’ and assume that the argument ends there.” Another » Continue Reading.
Scottish e-cig brand JAC Vapour is looking to take advantage of the growing support for e-cigarettes from Public Health England (PHE) by running an advertising promotion tied in to a national smoking cessation campaign.
Canada looks likely to regulate the sale of e-cigarettes as consumer products this autumn, ending years of uncertainty in which their legality has been disputed.
The U.S. Final Deeming Rule requires that the advertisements of all covered tobacco products, including e-cigarettes, bear an addictiveness warning label statement. For tobacco products that do not contain nicotine, there is an alternative warning label statement for packages and advertisements.
E-cigarette products are in the process of being regulated in Croatia, by transposing the Tobacco Product Directive (TPD), once the government is established. We expect the Draft Act on Restriction of the Use of Tobacco Products to be passed by the end of 2016, or early 2017. The delay in transposition led the European Commission (EC) to take an infringement decision against Croatia.
A new study published in The BMJ by researchers at University College London tests the contentious view that e-cigs, far from aiding in smoking cessation, can in fact undermine motivation to quit.
New research from Konstantinos Farsalinos could help to counter the misinformation on vaping that has become common in Malaysian policy circles.
Two new studies out of Canada come to different conclusions for the motivations of adults and children in taking up vaping.
A new study investigating young people’s perceptions of vaping and smoking has generated dramatic media headlines in the UK despite the tentative nature of the research findings.
The Cochrane systematic review of e-cigarettes and smoking cessation has just been updated, but many may wonder why it has been done now, as there is little new to say.
Taiwan may consider partial legalisation of nicotine e-cigs as part of a proposal to close loopholes in current laws, hoping to cut down on youth uptake.
Will e-cigarettes face the same battle of public opinion as the sugar substitute aspartame?
Nearly all smokers would be better off moving to lower-risk alternatives than giving up nicotine completely; taxing e-cigs has no rational justification; society should be prepared for more non-smokers to take up vaping; and tobacco control is often built on a “demonic possession theory”. These are among the conclusions of a provocative new paper published by the Institute of Economic Affairs and written by Carl V. Phillips.
A new study shows that youth e-cig users in the U.S. vape for flavours and not nicotine. The result has serious implications for future policy making decisions and justifications.
The effects of nicotine remain a major focus for scientific researchers investigating e-cigarettes, as three new studies indicate.
The majority of e-cig manufacturers and retailers have violated U.S. regulations about making health claims, according to a new study.
A majority of U.S. vapers will turn to the black market if new federal regulations result in their preferred products being withdrawn, research suggests.
A proposal has been submitted to Australian health authorities that would change the legal status of nicotine containing e-liquids if approved.
UK doctors would consider recommending e-cigarettes for patients looking to quit smoking, but they would not want to see them available on National Health Service (NHS) prescriptions.
Contents1 Executive summary2 Regulatory landscape3 National regulatory framework4 Age restrictions5 Labelling and packaging6 Retail channel restrictions (including cross-border and distance sales)7 Public usage8 Advertising and marketing9 Taxation10 Enforcement11 Case law12 Future developments There may be more recent ECigIntelligence reports on this territory. Please visit the home page for Russia or the advanced search page. Executive summary • E-cigarette » Continue Reading.
New advice from UK authorities advises consumers to switch from conventional smoking to vaping at home because of the reduced risk in fire as well as the health benefits for bystanders – particularly children.
A recent study and review by the National Institute for Public Health and the Environment in the Netherlands has brought to the fore questions regarding e-cigarette vapour testing methods and the possible health risks of passive vaping. The rationale for banning e-cigs in enclosed public places falls into two main camps: ‘renormalisation’ of smoking and possible safety concerns of passive » Continue Reading.
When the U.S. deeming regulations come into effect on 8th of August, the distribution of free e-liquid samples will be prohibited in the U.S. The U.S. Food and Drug Administration (FDA) justifies this ban as necessary for the protection of the public health but retailers believe it will undermine their profits without having a noticeable public health benefit. What challenges follow?
A new U.S. study claiming to show a link between youth e-cigarette use and tobacco smoking has not only been criticised by many scientists working in the field, but also demonstrates how misconceptions and misinformation on the topic of vaping spread through the media.
A new study from North Dakota says few vape stores comply with the state’s public vaping laws.
A scientific paper published today suggests that e-cigarettes could cut smoking-related deaths in the U.S. by more than 20%, based on a statistical model of how people smoke and vape.